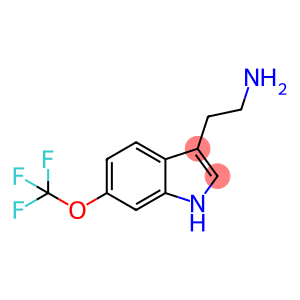
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride
CAS: 467451-81-6
Molecular Formula: C11H11F3N2O
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride - Names and Identifiers
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride - Physico-chemical Properties
| Molecular Formula | C11H11F3N2O |
| Molar Mass | 244.21 |
| Density | 1.37 |
| Boling Point | 335.6±37.0 °C(Predicted) |
| pKa | 16.11±0.30(Predicted) |
| Storage Condition | Room Temprature |
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride - Introduction
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride is a chemical compound with the formula C11H12F3N2O · HCl. The following is a detailed description of its nature, use, formulation and safety information:
Nature:
-Appearance: White to off-white crystalline powder
-Melting point: about 148-150°C
-Solubility: Soluble in water and ethanol
Use:
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride are commonly used in organic synthesis and pharmaceutical research. It can be used as a chemical reagent for the synthesis of a variety of compounds, especially compounds with aromatic and indole structures. In addition, it can also be used as a pharmaceutical intermediate in the synthesis of anti-tumor, anti-depression and anti-psychotic drugs.
Preparation Method:
The method for preparing 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride is to first synthesize 6-trifluoromethoxytryptamine, and then react with hydrochloric acid to form hydrochloride. Specific synthetic methods may vary depending on laboratory conditions and requirements.
Safety Information:
- 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride is a chemical substance and should be handled with care and follow safe operating procedures.
-It may be irritating to the eyes, skin and respiratory tract, so use appropriate protective measures, such as wearing protective glasses and gloves, to ensure good ventilation.
-Avoid inhalation or ingestion. If inhalation or accidental ingestion occurs, seek medical help immediately and provide proper emergency treatment.
-It should be stored in a dry, cool place, away from fire and oxidizing agents.
Please note that this is only a brief introduction. For specific applications and operations, reference should also be made to relevant literature and chemical laboratory guidance.
Nature:
-Appearance: White to off-white crystalline powder
-Melting point: about 148-150°C
-Solubility: Soluble in water and ethanol
Use:
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride are commonly used in organic synthesis and pharmaceutical research. It can be used as a chemical reagent for the synthesis of a variety of compounds, especially compounds with aromatic and indole structures. In addition, it can also be used as a pharmaceutical intermediate in the synthesis of anti-tumor, anti-depression and anti-psychotic drugs.
Preparation Method:
The method for preparing 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride is to first synthesize 6-trifluoromethoxytryptamine, and then react with hydrochloric acid to form hydrochloride. Specific synthetic methods may vary depending on laboratory conditions and requirements.
Safety Information:
- 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride is a chemical substance and should be handled with care and follow safe operating procedures.
-It may be irritating to the eyes, skin and respiratory tract, so use appropriate protective measures, such as wearing protective glasses and gloves, to ensure good ventilation.
-Avoid inhalation or ingestion. If inhalation or accidental ingestion occurs, seek medical help immediately and provide proper emergency treatment.
-It should be stored in a dry, cool place, away from fire and oxidizing agents.
Please note that this is only a brief introduction. For specific applications and operations, reference should also be made to relevant literature and chemical laboratory guidance.
Last Update:2024-04-09 20:49:11
Supplier List
Product Name: 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride Request for quotation
CAS: 467451-81-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 467451-81-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride Request for quotation
CAS: 467451-81-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 467451-81-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History
2-(6-(trifluoromethoxy)-1H-indol-3-yl)ethanamine hydrochloride
叔丁氧羰基-L-2,4-二氨基丁酸
PYRENE (13C3) SOLUTION 100UG/ML IN N-NONANE 1.2ML
Irloxacin
SODIUM AMIDE ( FLAKE)
(E)-3-(1-naphthyl)-N-[4-(1-pyrrolidinylsulfonyl)phenyl]-2-propenamide
邻三氟甲基苯乙烯(395-45-9)
Ethanol, 2-[[4-[(7-chloro-4-quinolinyl)amino]pentyl]amino]-, (R)-
65-42-9
695215-93-1
叔丁氧羰基-L-2,4-二氨基丁酸
PYRENE (13C3) SOLUTION 100UG/ML IN N-NONANE 1.2ML
Irloxacin
SODIUM AMIDE ( FLAKE)
(E)-3-(1-naphthyl)-N-[4-(1-pyrrolidinylsulfonyl)phenyl]-2-propenamide
邻三氟甲基苯乙烯(395-45-9)
Ethanol, 2-[[4-[(7-chloro-4-quinolinyl)amino]pentyl]amino]-, (R)-
65-42-9
695215-93-1
